
The following is a press release from Bayer regarding a voluntary recall. PATIENTS SHOULD NOT USE THIS PRODUCT!
Bayer is voluntarily recalling two lots of Kogenate® FS antihemophilic factor (recombinant) 2000 IU vials in the United States to the patient level. Certain vials from these two lots that were labeled as Kogenate FS actually contain the FVIII hemophilia A treatment, Jivi® antihemophilic factor (recombinant) PEGylated-aucl 3000 IU. The U.S. is the only country where affected products were distributed. We are working closely with the U.S. Food and Drug Administration to manage the recall and to minimize disruption to supply and inconvenience to patients. The affected lots, distributed from Feb. 5, 2019 to July 15, 2019 from Bayer’s distribution sites in Berkeley, Calif., and Shawnee, Kan., are listed below:
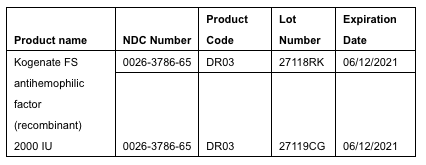

While the majority of the mislabeled vials in the affected lots were recovered, approximately 990 of these vials were released in the U.S. The associated Jivi batch was expired as of August 2018. However, all stability specifications of this expired Jivi batch had continued to be met, as of April 2019. We have voluntarily recalled both lots in the interest of patient safety, and to ensure that any potentially impacted product is removed from pharmacy shelves, and that patients and their healthcare providers are alerted. Importantly, vials of Kogenate FS that are not associated with the affected lot numbers (27118RK and 27119CG) are not impacted and can continue to be used without interruption. There are no lots of Jivi or Kovaltry® antihemophilic factor (recombinant) product affected by this recall.
Patient safety is Bayer’s highest priority. We are carefully monitoring for any complaints or adverse event reports that may be related to this recall. The company is issuing this notification after discovering the issue earlier this week. Following an immediate internal assessment, Bayer contacted the FDA to inform the agency of the situation.
Kogenate FS and Jivi are both medicines used to replace clotting factor (factor VIII or antihemophilic factor) that is missing in people with hemophilia A. Kogenate FS is approved to treat or control bleeding in adults and children with hemophilia A. Jivi is approved to treat and control bleeding in previously treated adults and adolescents (12 years of age and older) with hemophilia A.
Patients in possession of vials from the affected lot numbers should immediately stop using the product and contact their physician. In addition, patients should contact their pharmacy to return the affected product. Bayer is notifying its distributors in writing to check their stock immediately and to discontinue the distribution and use of any affected product. For distributors with questions regarding the recall process, please contact the Bayer Recall Coordinator, Inmar, at 855-707-7518.
Bayer is committed to providing our patients safe and effective therapy. We encourage healthcare providers and patients with questions to call our medical communications hotline with questions: 1-888-84-BAYER (1-888-842-2937).
We understand that this news may be concerning for patients with hemophilia A who depend on these medicines. Bayer is committed to providing the most up-to-date and accurate information to those who may be affected by this issue. Bayer takes its 30-year partnership with the hemophilia community very seriously, and we remain deeply dedicated to meeting the needs of patients living with this life-long disease.
Patients and healthcare providers are encouraged to report side effects or quality complaints of products to the FDA by visiting www.fda.gov/medwatch or calling 1-800-FDA-1088. For Bayer products, you can report these directly to Bayer at http://adversereactions.bayer.com.
Read Bayer’s Press Release Here.Â



