The following is sourced from Gilead. Read the update in its entirety “Gilead Warns of Counterfeit HIV Medication Being Distributed in the United States”. Click to view the original press release.
Foster City, Calif., August 5, 2021Â – Gilead Sciences has become aware of tampered and counterfeit versions of its once-daily single tablet HIV treatment regimen Biktarvy® (bictegravir 50 mg, emtricitabine 200 mg, and tenofovir alafenamide 25 mg tablets) and its HIV treatment and prevention medication Descovy® (emtricitabine 200 mg and tenofovir alafenamide 25 mg tablets) in circulation within U.S. drug distribution networks. Distributors not authorized by Gilead to sell Gilead-branded medicine have sold these counterfeits to pharmacies where genuine Gilead bottles have been tampered with a counterfeit foil induction seal or label and contain incorrect tablets. Working in communication with the U.S. Food and Drug Administration (FDA), Gilead has alerted potentially impacted pharmacies to investigate the potential for counterfeit or tampered Gilead medication sold by distributors not authorized by Gilead that may be within their recent supply and to remain vigilant to the potential for this to occur in the future. The authenticity and safety of Gilead-branded medicines can only be secure when obtained directly through Gilead’s authorized distributors.
Gilead continues to work closely with the FDA, pharmacies, and legal authorities to remove counterfeit and tampered medication from circulation and to prevent future distribution of these medications.
“The safety of individuals taking Gilead medication is always our first priority,” said Merdad Parsey, MD, PhD, Chief Medical Officer, Gilead Sciences. “We are taking aggressive action to ensure that healthcare providers and people who rely on our medicines can confidently distinguish authentic Gilead products from counterfeit drugs.”
Counterfeit and tampered medicines can bring serious and sometimes life-threatening health risks to individuals. These medicines are not equivalent in quality, safety, and/or efficacy to genuine medicines. They often do not contain the correct medicine or amount of active ingredient and may also contain impurities. Additionally, these medicines may be produced in unsafe manufacturing conditions and travel through insecure supply chains.
The following elements can help ensure dispensed Biktarvy and Descovy are authentic:
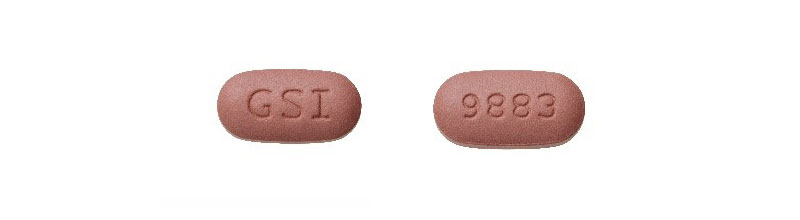
- Authentic Biktarvy tablets are purplish-brown, capsule-shaped pills with “9883” on one side and “GSI” on the other.
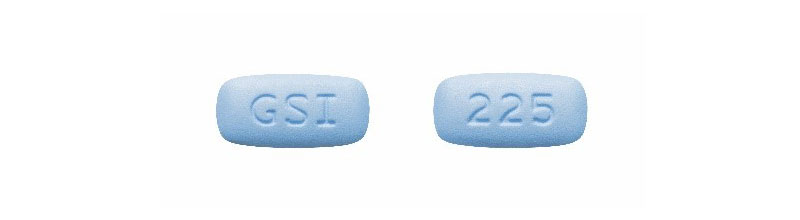
- Authentic Descovy tablets are blue, rectangular pills with “225” on one side and “GSI” on the other.
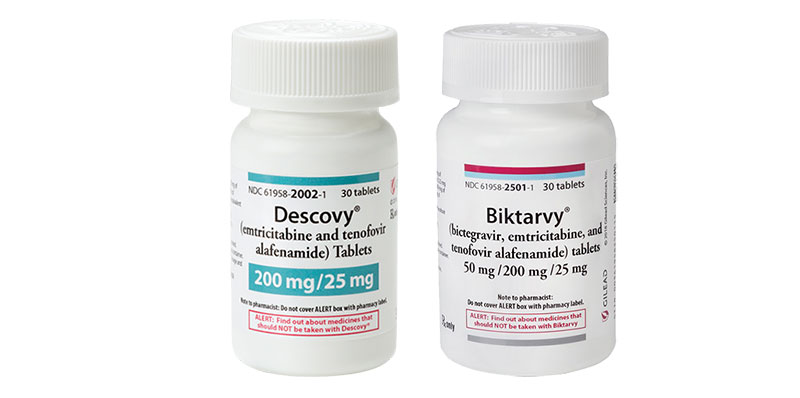
- FDA requires that Biktarvy and Descovy are dispensed in original packaging. Confirm your dispensed 30-count bottle of Biktarvy or Descovy are received in bottles that are white plastic, with white plastic caps, and Gilead-branded labels.
- Confirm your Biktarvy and Descovy were dispensed from a pharmacy that sources Gilead medicine directly from Gilead authorized distributors. A list of Gilead’s authorized distributors can be found here.
Individuals who believe they have been dispensed a counterfeit and/or tampered Gilead medication should immediately report the medicine to their doctor and pharmacy and Gilead Product Quality Complaints (1-800-445-3235, Option #2; QualityComplaints@gilead.com). If an individual is experiencing any side effects that may be related to a Gilead medication or to the use of a counterfeit drug, that person should immediately contact their healthcare provider and is additionally encouraged to report the event to FDA’s MedWatch Safety Information and Adverse Event Reporting Program (1-800-FDA-1088 or www.fda.gov/medwatch) or Gilead (1-800-445-3235, Option #3). Websites selling counterfeit and/or tampered medicines may be reported to the FDA. Healthcare professionals are encouraged to report sales solicitation of suspect drug products by emailing DrugSupplyChainIntegrity@fda.hhs.gov and by calling the FDA’s Office of Criminal Investigations (OCI) at 800-551-3989 or a local OCI field office.
Additional information about Biktarvy
What is BIKTARVY?
BIKTARVY is a complete 1-pill, once-a-day prescription medicine used to treat HIV-1 in adults and children who weigh at least 55 pounds. It can either be used in people who have never taken HIV-1 medicines before, or people who are replacing their current HIV-1 medicines and whose healthcare provider determines they meet certain requirements. BIKTARVY does not cure HIV-1 or AIDS. HIV-1 is the virus that causes AIDS.
What is the most important information I should know about BIKTARVY?
BIKTARVY may cause serious side effects:
- Worsening of hepatitis B (HBV) infection. Your healthcare provider will test you for HBV. If you have both HIV-1 and HBV and stop taking BIKTARVY, your HBV may suddenly get worse. Do not stop taking BIKTARVY without first talking to your healthcare provider, as they will need to monitor your health, and may give you HBV medicine.
For additional Important Safety Information about Biktarvy, including important warnings, please talk to your healthcare provider and visit www.Biktarvy.com.
Additional information about Descovy for the treatment of HIV-1 infection
What is DESCOVY for HIV-1 treatment?
DESCOVY is a prescription medicine that is used together with other HIV-1 medicines to treat HIV-1 in people who weigh at least 77 lbs (35kg). DESCOVY combines 2 medicines into 1 pill taken once a day. Because DESCOVY by itself is not a complete treatment for HIV-1, it must be used together with other HIV-1 medicines. DESCOVY does not cure HIV-1 or AIDS. HIV-1 is the virus that causes AIDS.
What is the most important information I should know about DESCOVY?
DESCOVY may cause serious side effects:
- Worsening of hepatitis B (HBV) infection. Your healthcare provider will test you for HBV. If you have both HIV-1 and HBV and stop taking DESCOVY, your HBV may suddenly get worse. Do not stop taking DESCOVY without first talking to your healthcare provider, as they will need to monitor your health or give you HBV medicine.
For additional Important Safety Information about DESCOVY for the treatment of HIV-1 infection, including important warnings, please talk to your healthcare provider and visit www.Descovy.com/treatment.
Additional information about Descovy for the prevention of HIV-1 infection
What is DESCOVY for PrEP?
DESCOVY for PrEP (pre-exposure prophylaxis) is a once-daily prescription medicine for adults and adolescents at risk of HIV. It helps lower the chances of getting HIV through sex.
DESCOVY for PrEP is not for everyone:
- It is not for use in people assigned female at birth who are at risk of getting HIV from vaginal sex, because its effectiveness has not been studied.
- You must be HIV-negative before and while taking DESCOVY for PrEP.
What is the most important information I should know about DESCOVY for PrEP?
Before and while taking DESCOVY for PrEP:
- You must be HIV-negative before you start and while taking DESCOVY for PrEP. You must get tested for HIV-1 immediately before and at least every 3 months while taking DESCOVY. If you think you were exposed to HIV-1, tell your healthcare provider right away. They may want to do more tests to confirm that you are still HIV-negative.
- Many HIV-1 tests can miss HIV-1 infection in a person who has recently become infected. Tell your healthcare provider if you had a flu-like illness within the last month before starting or while taking DESCOVY. Symptoms of new HIV-1 infection include tiredness, fever, joint or muscle aches, headache, sore throat, vomiting, diarrhea, rash, night sweats, and/or enlarged lymph nodes in the neck or groin.
- DESCOVY by itself is not a complete treatment for HIV-1. Do not take DESCOVY for PrEP unless you are confirmed to be HIV-1 negative.
- DESCOVY does not prevent other sexually transmitted infections (STIs). Practice safer sex by using a latex or polyurethane condom to reduce the risk of getting STIs.
- To further help reduce your risk of getting HIV-1:
- Do not miss any doses of DESCOVY. Missing doses may increase your risk of getting HIV-1.
- Know your HIV status and the HIV status of your partners. If your partner is living with HIV, your risk of getting HIV is lower if your partner consistently takes HIV treatment every day.
- Get tested for other STIs. Some STIs make it easier for HIV-1 to infect you.
- Talk to your healthcare provider about all the ways to help reduce HIV risk.
DESCOVY may cause serious side effects:
- Worsening of hepatitis B (HBV) infection. Your healthcare provider will test you for HBV. If you have both HIV-1 and HBV and stop taking DESCOVY, your HBV may suddenly get worse. Do not stop taking DESCOVY without first talking to your healthcare provider, as they will need to monitor your health or give you HBV medicine.
For additional Important Safety Information about Descovy for PrEP, including important warnings, please talk to your healthcare provider and visit www.Descovy.com/prep. Â
About Gilead Sciences
Gilead Sciences, Inc. is a biopharmaceutical company that has pursued and achieved breakthroughs in medicine for more than three decades, with the goal of creating a healthier world for all people. The company is committed to advancing innovative medicines to prevent and treat life-threatening diseases, including HIV, viral hepatitis and cancer. Gilead operates in more than 35 countries worldwide, with headquarters in Foster City, California.
For more than 30 years, Gilead has been a leading innovator in the field of HIV, driving advances in treatment, prevention, and cure research. Today, millions of people living with HIV globally receive antiretroviral therapy provided by Gilead or one of the company’s manufacturing partners. Â
U.S. Prescribing Information for Biktarvy and Descovy, including BOXED WARNINGS, is available at www.gilead.com.
Gilead, Biktarvy, Descovy, and Descovy for PrEP, “9883”, Biktarvy “9883” Tablet Design, and the Blue Tablet Design are registered trademarks of Gilead Sciences, Inc., or its related companies.
For more information about Gilead, please visit the company’s website at www.gilead.com, follow Gilead on Twitter (@Gilead Sciences) or call Gilead Public Affairs at 1-800-GILEAD-5 or 1-650-574-3000.



